 Sete Orthopedic Implants
Sete Orthopedic Implants
 Sete Orthopedic Implants
Sete Orthopedic Implants
Shanghai Sete Technology — 30+ years supplying full-range orthopedic solutions with complete OEM & ODM capabilities backed by a world-class supply chain.

Shanghai Sete Technology Co., Ltd. is a leading manufacturer and supplier of premium orthopedic implants, built on a complete, vertically integrated supply chain. From raw material selection through precision CNC machining, polishing, ultrasonic cleaning, and final inspection, every product is engineered to the highest clinical standards.
With over 30 years of industry experience and ISO & CE certifications, we deliver full-range orthopedic product series — including artificial knee joints, hip replacement systems, ceramic femoral heads, and custom OEM/ODM solutions — to distributors, hospitals, and surgical centers across more than 50 countries.
Our mission is simple: provide world-class orthopedic products with responsive service, competitive pricing, and zero minimum order quantity — empowering our partners to grow confidently in their markets.
From total knee replacement systems to ceramic femoral heads, every Sete implant is manufactured under strict quality protocols to ensure optimal patient outcomes and surgical reliability.

Our SKII CR Femoral Condyle system delivers anatomically accurate geometry for natural knee kinematics, precise bone resection, and long-term implant stability — available in multiple sizes with full surgical instrument sets.

Engineered from high-purity alumina ceramic, our THA Femoral Head offers exceptional hardness, ultra-low wear rates, and outstanding biocompatibility — ideal for active patients requiring long-lasting total hip arthroplasty solutions.
Our ISO-certified manufacturing campus integrates advanced CNC machining centers, precision polishing lines, ultrasonic cleaning systems, and a fully equipped QC laboratory — ensuring every implant meets international surgical standards before it leaves our facility.
Download Product CatalogEvery orthopedic implant undergoes a rigorous, fully traceable production process — from certified raw material sourcing to final sterile packaging — ensuring consistent quality and regulatory compliance at every stage.






We go beyond product supply — we become a strategic partner invested in your growth, your surgeons' success, and your patients' outcomes.
Prompt response to every enquiry within 2 hours — because your time and your customers' needs cannot wait.
Competitive prices with zero minimum order requirement, empowering distributors to test quality and market demand risk-free.
All products are manufactured under ISO-certified processes and carry CE certification, meeting international regulatory requirements.
Experienced logistics team ensures efficient, compliant, and on-time delivery to distributors worldwide with full export documentation.
We provide surgical technique training workshops in China and send experienced surgeons to support live surgery demonstrations for distributors.
We actively sponsor advertisement campaigns and international medical exhibitions to support our exclusive distributors' market development.
Favorable and flexible payment terms are available for our reliable exclusive distributors, building long-term partnerships based on mutual trust.
Our engineering team continuously tracks the latest surgical techniques and design trends, developing new products and improving existing lines to stay ahead of market needs.
Our certifications reflect our unwavering commitment to product safety, manufacturing excellence, and global regulatory compliance — giving distributors and surgeons the confidence they need.
Our manufacturing facility operates under a fully implemented ISO Quality Management System, ensuring every process — from procurement to delivery — meets internationally recognized standards for medical device production.
All Sete orthopedic implants carry the CE Mark, confirming full compliance with European Medical Device Regulations and providing our global partners with unrestricted access to EU and associated markets.
With over 30 years of manufacturing expertise, we offer comprehensive OEM and ODM services — from design customization and material selection to private labeling and regulatory documentation support for your market.
A visual walkthrough of the precision manufacturing journey that transforms certified raw materials into world-class orthopedic implants.




Our partners across Europe, Asia, and the Middle East consistently highlight our product quality, responsive support, and commitment to their long-term success.
"Sete's knee implant systems have consistently delivered excellent surgical outcomes for our patients. The quality is on par with leading global brands, but with far more competitive pricing and genuinely responsive support."
"What sets Sete apart is their willingness to support us beyond just product supply. The surgical training workshops in China were invaluable for our team, and their 2-hour response policy is genuinely upheld."
"We started with a small trial order — no MOQ pressure at all. The product quality passed our internal testing immediately. We are now their exclusive distributor in our region and couldn't be more satisfied."
Join distributors and surgical centers in 50+ countries who trust Sete Medical for premium orthopedic implants, responsive service, and genuine partnership support.
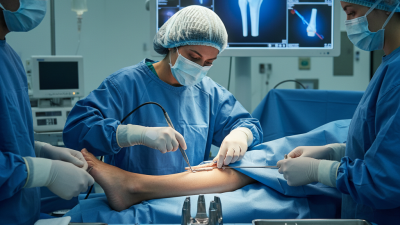
In the field of orthopedic surgery, understanding "how are orthopedic implants inserted" is crucial for successful outcomes. Dr. Emily Carter, an
Author: Isabella Date: 2026-05-31
Learn More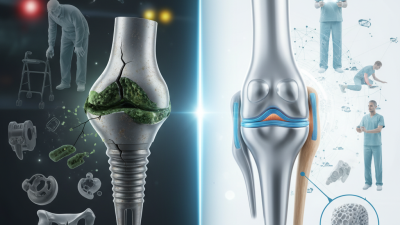
Orthopedic implants play a crucial role in restoring mobility and alleviating pain for countless patients. However, the alarming rate of implant
Author: Sienna Date: 2026-05-28
Learn More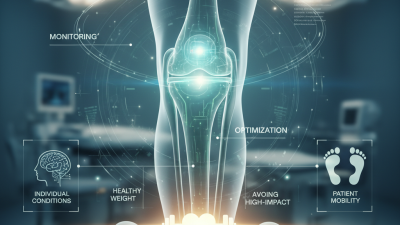
In the field of orthopedic medicine, the longevity of implants is a crucial concern. Dr. Emily Thompson, a renowned orthopedic surgeon, once stated,
Author: Amelia Date: 2026-05-25
Learn More