

 Sete Orthopedic Implants
Sete Orthopedic Implants










| Products Name | NO. | Specification |
|---|---|---|
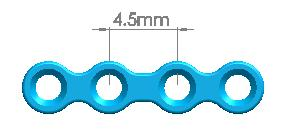
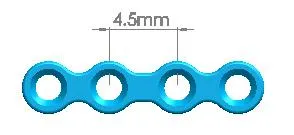
| Straight Maxillofacial Plate, Type-III | 1500-0110 | 4 holes (Interspace: 4.5mm) |
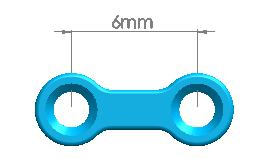
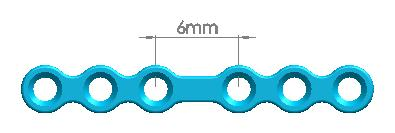
| 1500-0111 | 4 holes (Interspace: 6.0mm) | |
| 1500-0112 | 4 holes (Interspace: 7.5mm) | |
| 1500-0113 | 4 holes (Interspace: 9.0mm) | |
| 1500-0114 | 4 holes (Interspace: 12mm) | |
| Use: 1.5mm Self-tapping Screw / 1.5mm Self-drilling Screw | ||

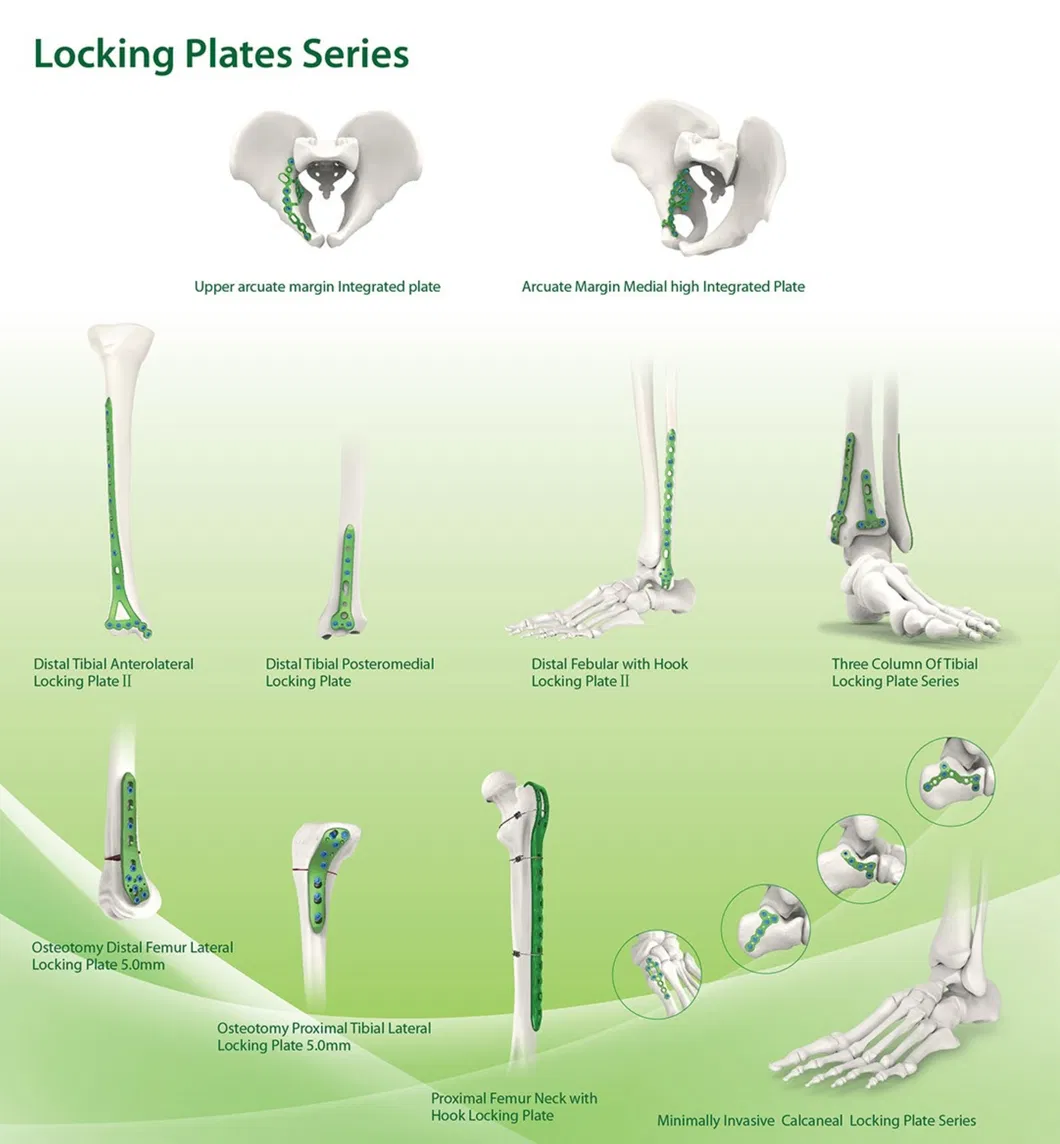






The enterprise was established in 1958 and is dedicated to the development, manufacture, and sales of orthopedic implants and instruments. As a key entity in the medical apparatus industry, the product line covers spine pedicle screws, interlocking nails, locking plates/screws, trauma plates, cannulated screws, external fixators, and surgical instruments.
All medical apparatuses have passed rigorous approval and authentication by national and international regulatory offices. Products are characterized by multi-varieties and complete specifications, featuring laser anti-fake marks and material codes for quality tracking.
The facility utilizes advanced equipment, including imported CNC machines, high-precision digital control systems, and a 100,000 Grade axenic purification chamber to ensure high-quality production standards. The company was among the first to achieve ISO 9001 quality system certification and has received numerous awards for science and technology innovation.
