


 Sete Orthopedic Implants
Sete Orthopedic Implants






The Interface Screw is designed for use in ACL reconstruction surgeries to secure tendon or ligament grafts in the femoral and tibial bone tunnels. It provides a strong and reliable fixation by compressing the tendon or ligament graft directly against the bone tunnel wall.
The screw's unique mechanism ensures optimal stability and a firm attachment between the soft tissue graft and the bone. The system includes a conical screw for easy insertion and a protective sheath that shields the soft tissue, reducing the risk of graft displacement or screw breakage. Made from PEEK (Polyetheretherketone), it mimics the elasticity of natural bone, ensuring superior performance during healing.
The tapered shape ensures easy insertion, with a head diameter larger than the tip to facilitate precise placement.
High-strength design prevents damage during insertion and enhances holding power under torque.
A circular sheath protects the tendon and ligament from being cut, minimizing damage risk during the surgical process.
External clamp blocks ensure the graft remains securely in place, even under significant external pressure.
Offers excellent biocompatibility and elasticity similar to natural bone, promoting optimal integration.
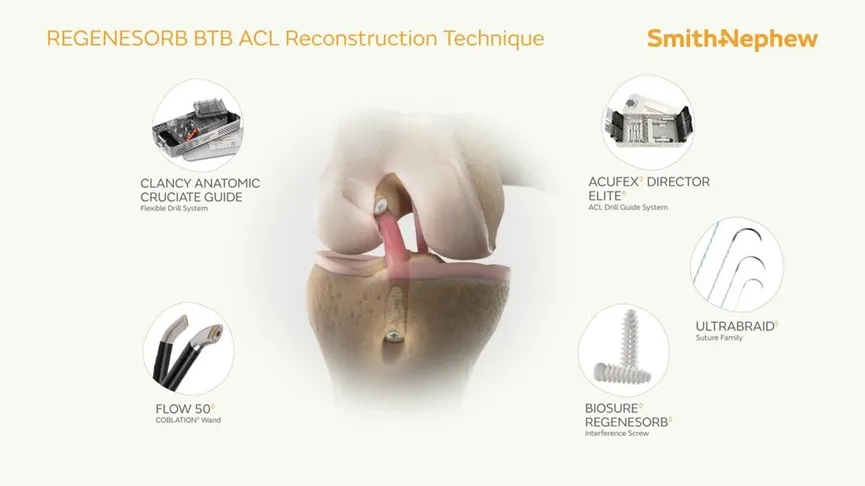

This Interface Screw System is primarily used for ACL reconstruction surgeries, where it provides secure fixation for soft tissue grafts or bone-tendon grafts in the knee joint, ensuring effective healing and recovery.


Standardized Orthopedic Solutions
We are a leading manufacturer of standardized orthopedic implants and instruments, serving global distributors and importers. Our full product range covers trauma, spine, joints, sports medicine, and CMF.
With in-house R&D, certified laboratories, and modern production lines, we provide high-quality surgical solutions designed for reliable performance and optimal clinical outcomes. We focus on scalable manufacturing to meet high-volume distribution needs.