Q: What are the primary materials used in this system?
A: The system utilizes high-grade materials including TC4 ELI titanium alloy, CoCrMo alloy (Co28Cr6Mo), and pure titanium (TA2G) to ensure biocompatibility and mechanical strength.
Q: Are your manufacturing processes certified?
A: Yes, we hold EN ISO 13485 certification, ensuring that all medical devices meet stringent global safety and performance standards.
Q: What is the minimum order requirement?
A: Our standard minimum order value is US$1000. However, sample orders are available at cost for evaluation and market testing.
Q: Do you support custom product development?
A: Yes, we accommodate custom orders. By providing pictures or samples, we can work with you to create specific orthopedic solutions, though lead times may vary.
Q: How do you handle international shipping?
A: We primarily use professional courier services. Shipping costs are calculated based on destination, package weight, and size, with detailed quotes provided upon request.
Q: Is inventory readily available for catalogue items?
A: We maintain stock for most catalogue items with regular monthly production cycles. For real-time inventory status, please consult our sales team.


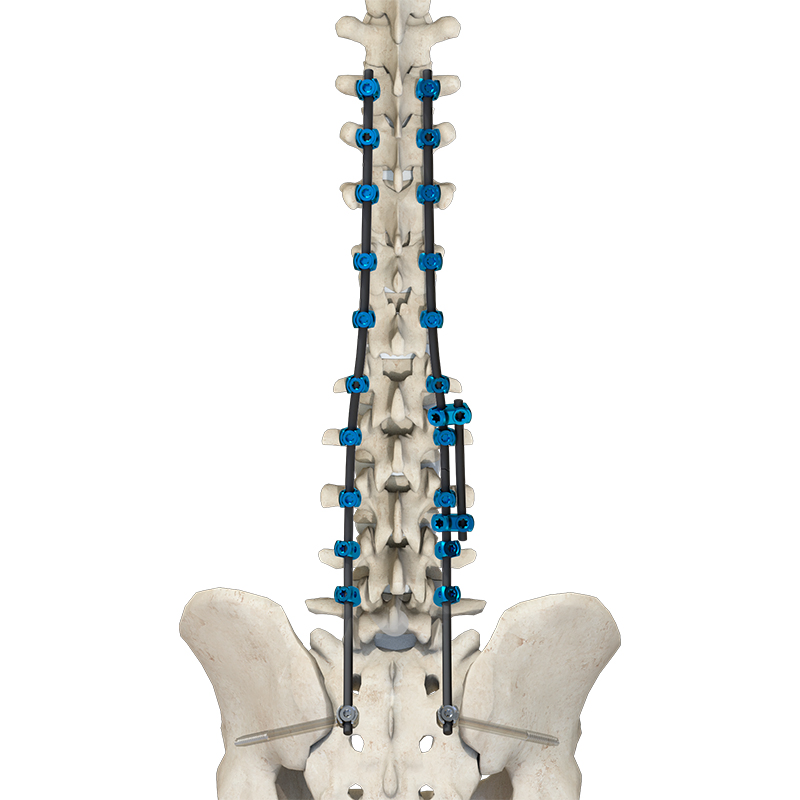
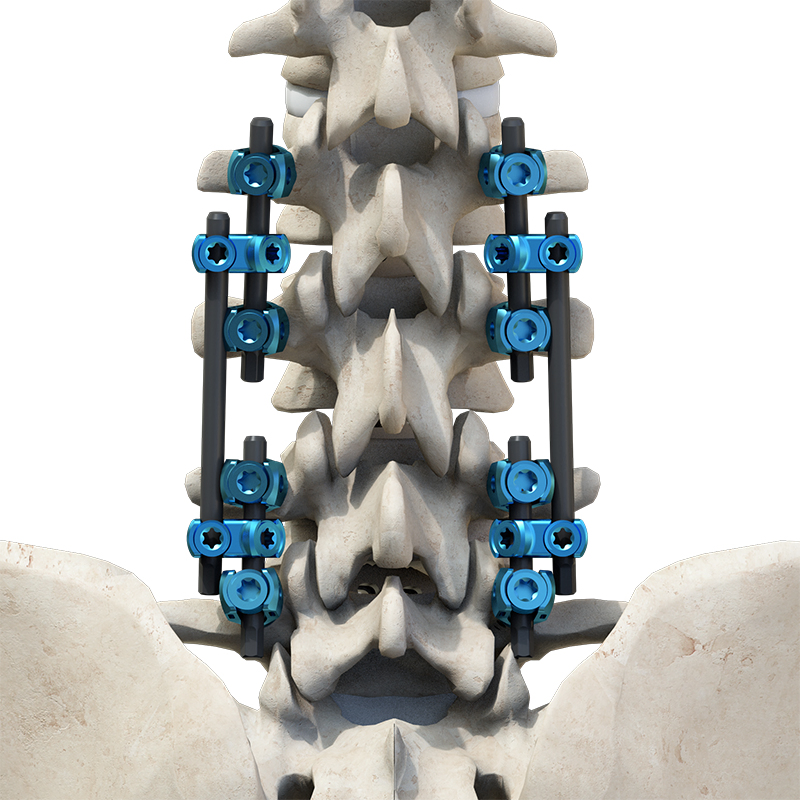
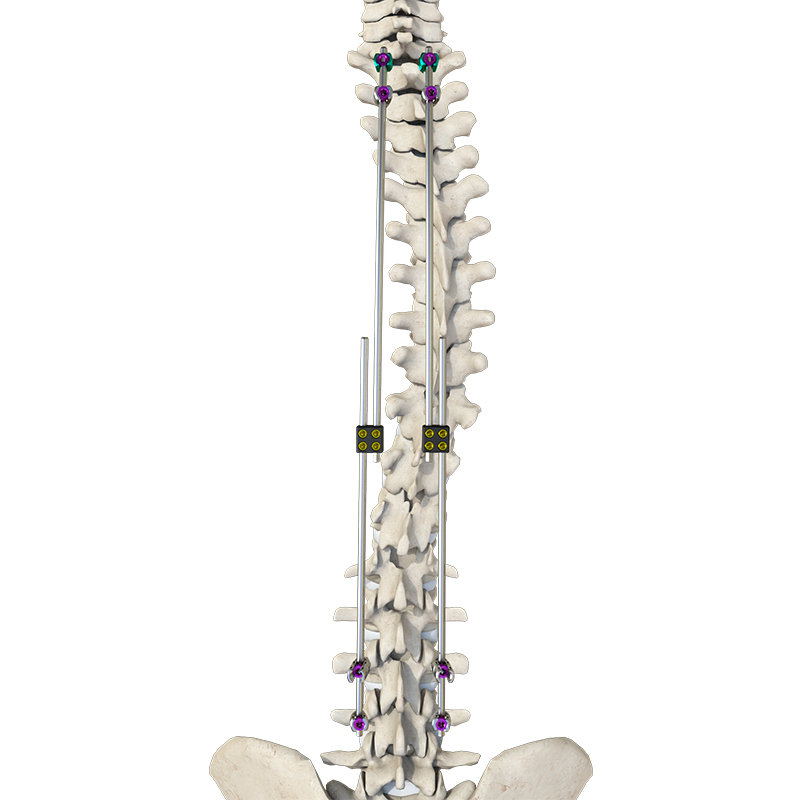
 Sete Orthopedic Implants
Sete Orthopedic Implants












