
1 / 5
 Sete Orthopedic Implants
Sete Orthopedic Implants












| Model Number | Spec.(mm) |
|---|---|
| 1072001 | S |
| M | |
| L |

Professional Orthopedic Manufacturer You Can Trust
We are a leading manufacturer of standardized orthopedic implants and instruments for global distributors and importers.
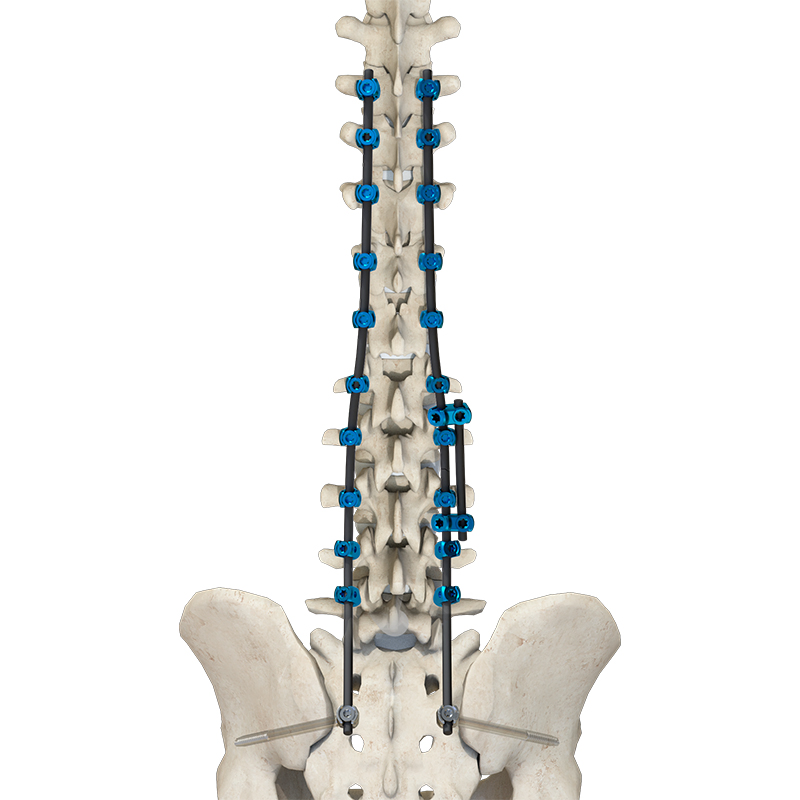
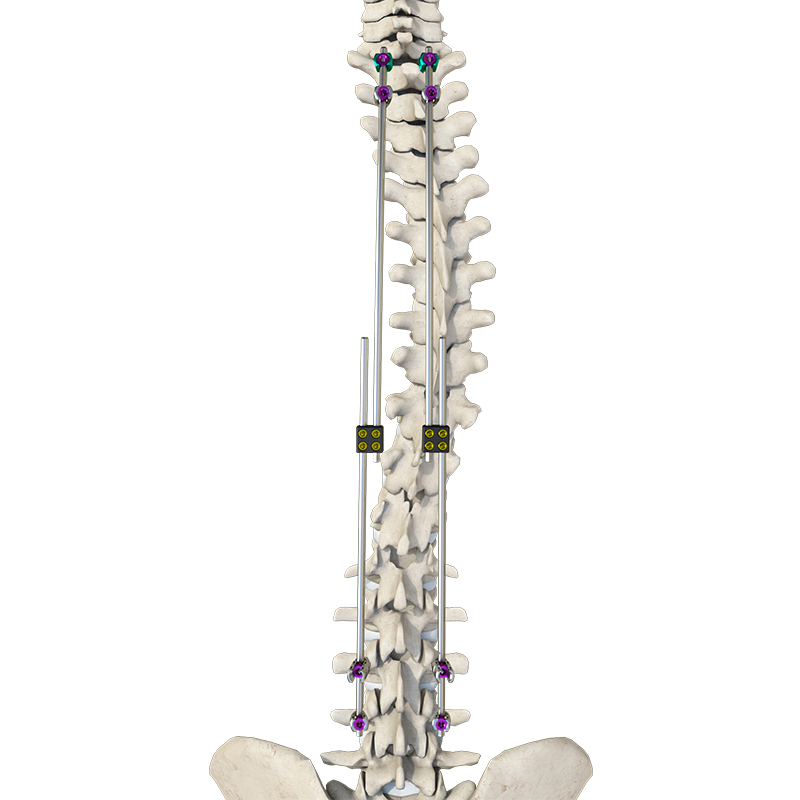
Our full product range covers trauma, spine, joints, sports medicine, and CMF — all supported by complete surgical instrument sets. Our products are designed for frequent orders, low MOQ, and fast turnaround, making them ideal for emerging markets and high-volume distribution.
With in-house R&D, a certified lab, and modern production lines, we also support OEM/ODM services for partners who need private label or custom solutions. We focus on reliable, scalable manufacturing so you can focus on growing your market.