




 Sete Orthopedic Implants
Sete Orthopedic Implants











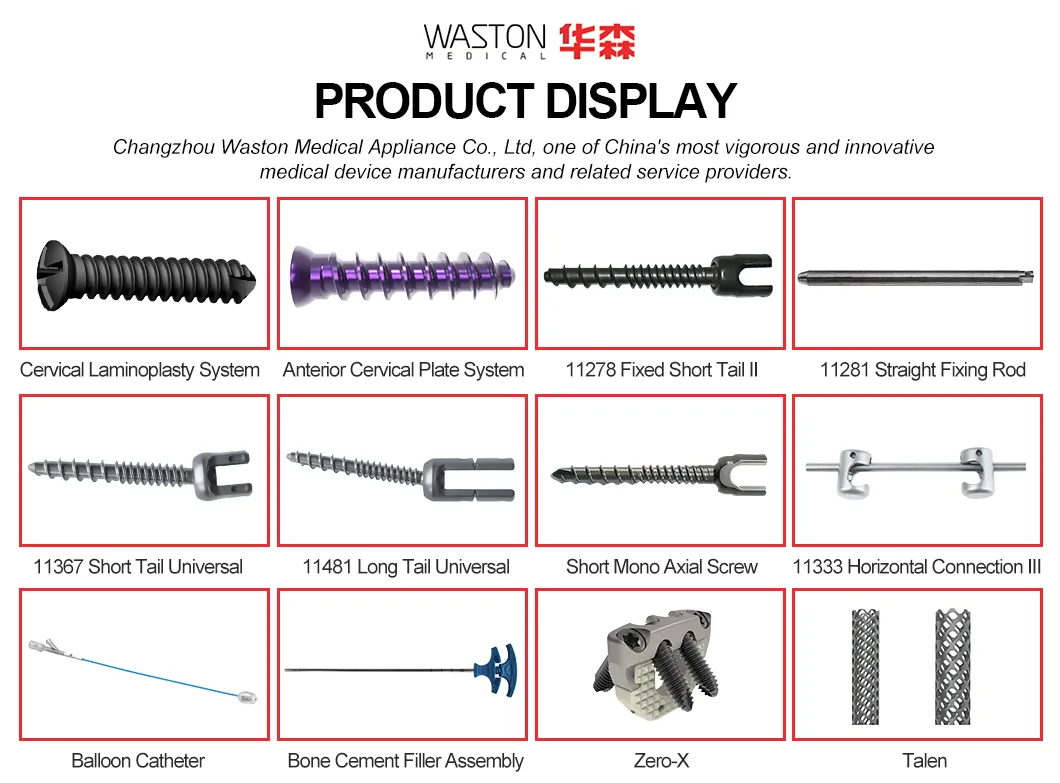
| PRODUCT NAME | PRODUCT CODE | DIA | DATA |
|---|---|---|---|
| NEULEN Laminoplasty Plate | 11101012 | / | H=12 |
| 11101014 | / | H=14 | |
| 11101016 | / | H=16 | |
| NEULEN Screw (Self-tapping) | 10511306 | φ2.5 | L=6 |
| 10511308 | φ2.5 | L=8 | |
| 10511310 | φ2.5 | L=10 |

As a national high-tech enterprise, R&D investment accounts for over 8% of total sales. We focus on transforming clinical experience into innovative medical achievements, pioneering technologies in Rev Drill systems, Three-Row-Staplers, and Sternal Fixation.
Operating under ISO13485 and CE MDD 93/42/EEC directives, our facility utilizes German, American, and Japanese imported machining centers and automatic lathes to ensure first-class quality. Our testing center includes biomechanical and physical-chemical laboratories for rigorous raw material and product performance analysis.

Our orthopaedic solutions are exported to numerous countries, including Columbia, France, Turkey, Indonesia, Mexico, Russia, and Thailand. We also serve as a major OEM supplier for some of the world's largest players in the medical device industry.




Yes, we support customized designs based on specific surgical requirements or drawings.
We hold CE and ISO certifications. Our Spinal System also carries FDA approval.
Yes, we welcome sample orders to test our product quality and service before placing bulk orders.
Samples are typically delivered within 7 days, while standard orders are processed within 25 days.
For any defective items, please provide photographic evidence for confirmation, and we will arrange for replacements.
Generally, we maintain stock for listed products, though availability depends on current sales conditions. Please contact our sales team for accurate inventory data.