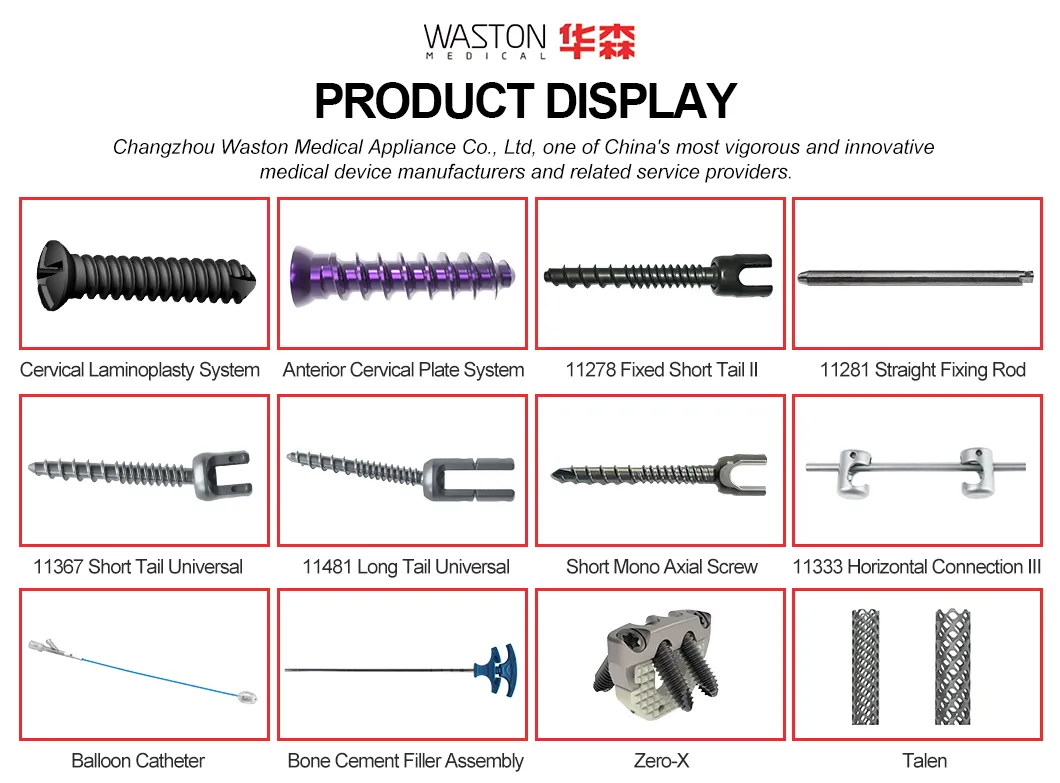
Basic Information
Material
Pure Titanium/Titanium Alloy/Stainless Steel
Certification
CE, ISO, SGS
Package
Non-Sterile Package, Paper Plastic Roll Bag
Product Introduction
★ Key Features
✔ Immediate lamina supporting in surgery, best maintain the canal plasty expanded.
✔ The lower dentate supporting plate provides a more stable supporting force.
✔ Arc openings provide better inclusion.
✔ High plasticity allows for slight moulding during operations.
★ Advantages
✚ Provides stable support for vertebral body decompression.
✚ Four-hole design meets diverse surgical requirements.
✚ Unique angle design enhances vertebral body stability.
Product Parameters & Specifications
| PRODUCT NAME |
PRODUCT CODE |
DIA |
DATA |
| Laminoplasty Plate |
11101012 |
/ |
H=12 |
| 11101014 |
/ |
H=14 |
| 11101016 |
/ |
H=16 |
| Screw (Cross self-tapping) |
10511306 |
φ2.5 |
L=6 |
| 10511308 |
L=8 |
| 10511310 |
L=10 |
Product Recommendation Display
Technology & Manufacturing

As a national high-tech enterprise, R&D investment accounts for over 8% of total sales. We focus on transforming clinical experience into innovative solutions. We pioneer revolutionary technology in Rev Drill systems, Three-Row-Staplers, Sternal Fixation, and Rib plates.

Operating strictly under the ISO13485 quality system and CE MDD 93/42/EEC directives. Our facility features German, American, and Japanese-imported machining centers and automatic lathes to ensure first-class quality. Our testing center includes biomechanical and physical-chemical laboratories.
Global Exhibition & OEM
Our products are exported to numerous countries including Columbia, France, Turkey, Indonesia, Mexico, Russia, and Thailand. We also serve as a major OEM supplier for leading global players in the medical device industry.
Frequently Asked Questions
1. Q: Is customized design available for these implants?
A: Yes, we provide professional customization based on drawings or samples provided by the customer.
2. Q: What is your return policy for defective goods?
A: For any defective goods, please provide photographic evidence for confirmation, and we will arrange for a suitable replacement.
3. Q: Can I request samples for quality and market testing?
A: Yes, we welcome sample orders to test our product quality and service before proceeding with bulk orders.
4. Q: What certificates do you hold for these medical systems?
A: We hold CE and ISO certifications. For Spinal Systems, we also possess FDA registration.
5. Q: What is the typical delivery timeframe?
A: For samples, it is typically within 7 days. For standard production orders, the delivery time is approximately 25 days.
6. Q: Do you maintain inventory for regular items?
A: We generally stock listed products. However, availability depends on current sales conditions; please contact our sales team for real-time inventory information.


 Sete Orthopedic Implants
Sete Orthopedic Implants