1 / 5
 Sete Orthopedic Implants
Sete Orthopedic Implants










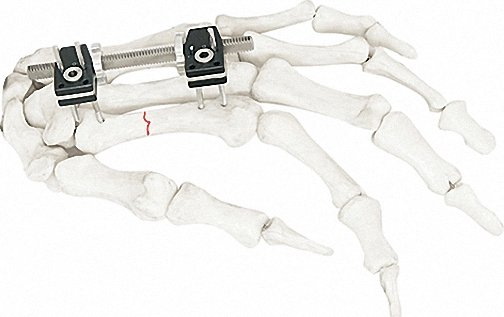
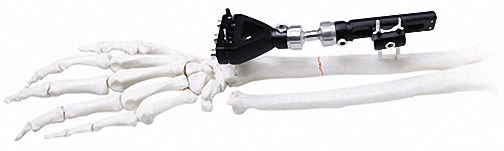
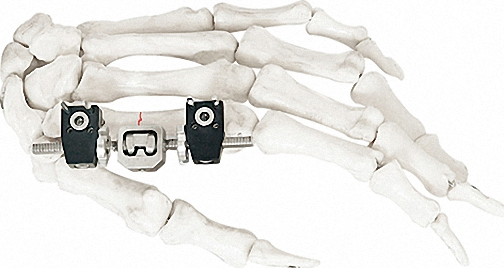
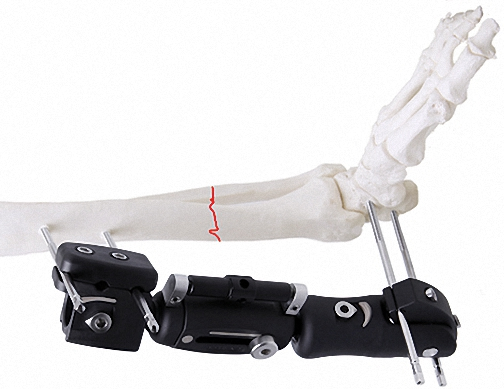
External Fixator -- The Pinnacle of Modular Fixation Systems
The ultimate solution in bone fixation, setting a new standard in the industry with unparalleled stability and modular flexibility.



| Component Name | Specification | Qty |
|---|---|---|
| Independent Coupling | φ6*φ14(1.5-2.0) | 10 |
| Needle Clip | φ4*φ9 (φ1.0) | 5 |
| Cone Washer | φ4*φ9 | 10 |
| Connection Plate | φ4*20 - φ4*50 | 4/each |
| Fixator (Hinge) | M4*20mm | 4 |
| Straight Threaded Rod | M3/M4 Various Sizes | 2-8 |
| Bone Screws | 1.0/1.2/1.5*150 | 8-20 |
| Instrument Set | Standard | 1 |



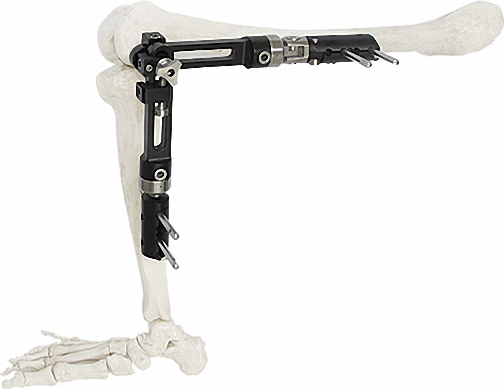
Ring Fixator (Tibia Femur)

2/3 Ring Fixator

Ankle Joint Fixator


