Product Description
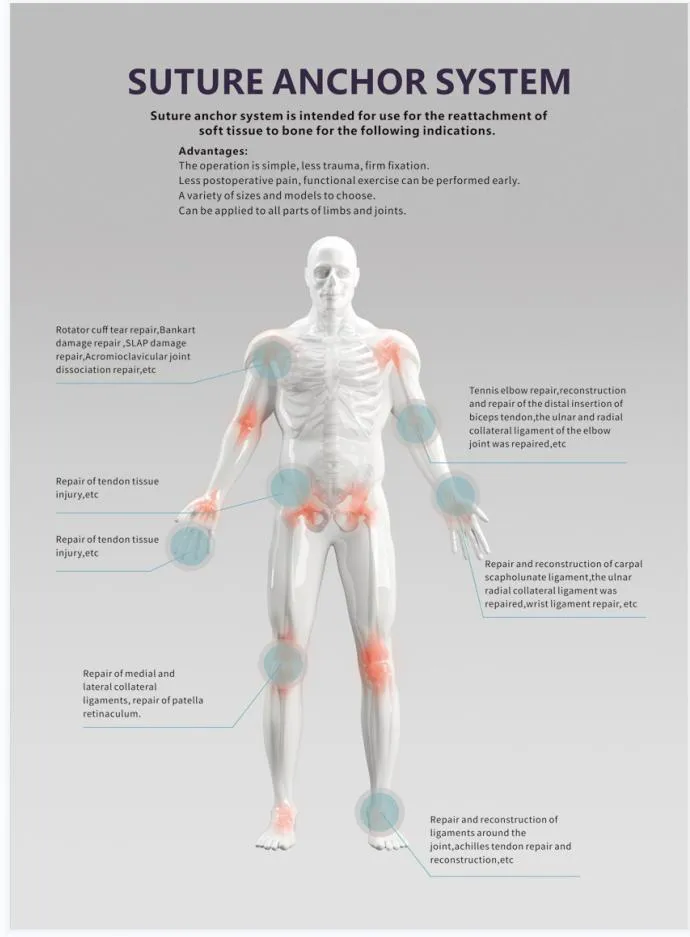
Sports Medicine Surgery for Repair Restruction Rotator Cuff Tendon Suture Anchor.
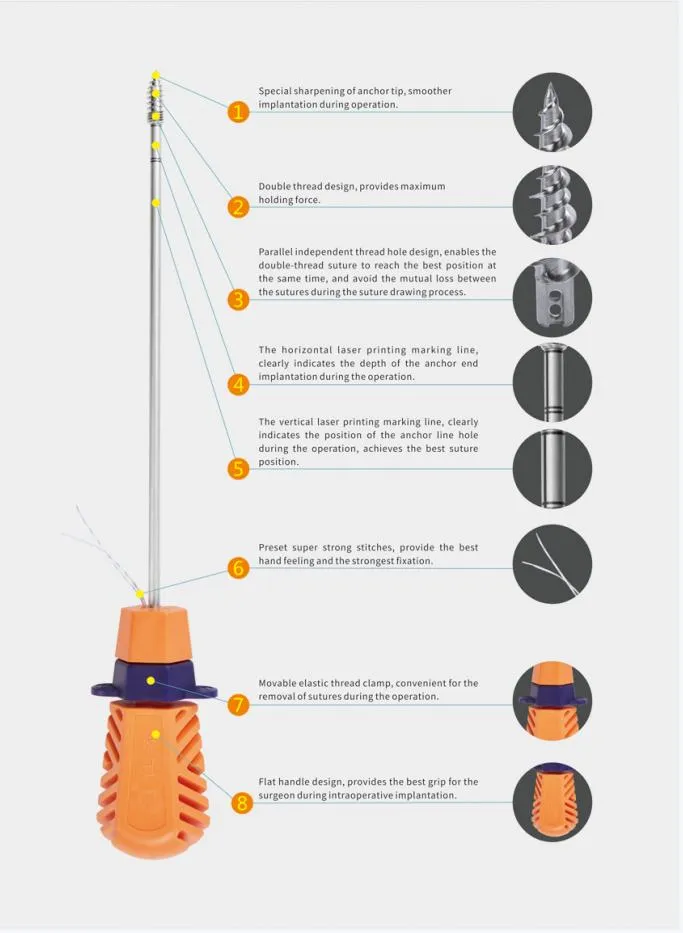
The Suture Anchor System consists of anchor, nonabsorbable suture and inserter. Anchor is made of Ti6Al4V alloy, which meets the requirement of ISO5832-3:2016. Suture is made of ultra high molecular weight polyethylene material without coating, it is woven from several UHMWPE yarn, which meets the requirement of ASTM F2848-17. Blue suture is from white ultra high molecular weight polyethylene, dyed with blue color material (FDA approved). Part of inserter contacting human body is made of stainless steel conforming ASTM F899-12b. Handle of inserter is made of polycarbonate and ABS material.
Frequently Asked Questions
What materials are used for the Suture Anchor?
The anchor is constructed from high-quality Ti6Al4V titanium alloy, which strictly adheres to ISO5832-3:2016 standards for surgical implants.
What is the composition of the suture?
The suture is made from ultra-high molecular weight polyethylene (UHMWPE) yarn. It is non-absorbable and meets the ASTM F2848-17 requirements.
How is the product sterilized?
The system is EO (Ethylene Oxide) sterilized and provided in individual sterile blister packaging to ensure maximum safety for orthopedic procedures.
What specific surgical applications is this system used for?
It is primarily designed for Sports Medicine and Orthopedic surgery, specifically for repairing and reconstructing rotator cuff tendons by connecting soft tissue to bone.
Does the product carry international certifications?
Yes, the suture anchor system is certified with CE and ISO13485, classifying it as a Class II medical instrument.
Are samples available for evaluation?
Yes, samples can be provided for professional clinical evaluation upon request.





 Sete Orthopedic Implants
Sete Orthopedic Implants