


 Sete Orthopedic Implants
Sete Orthopedic Implants











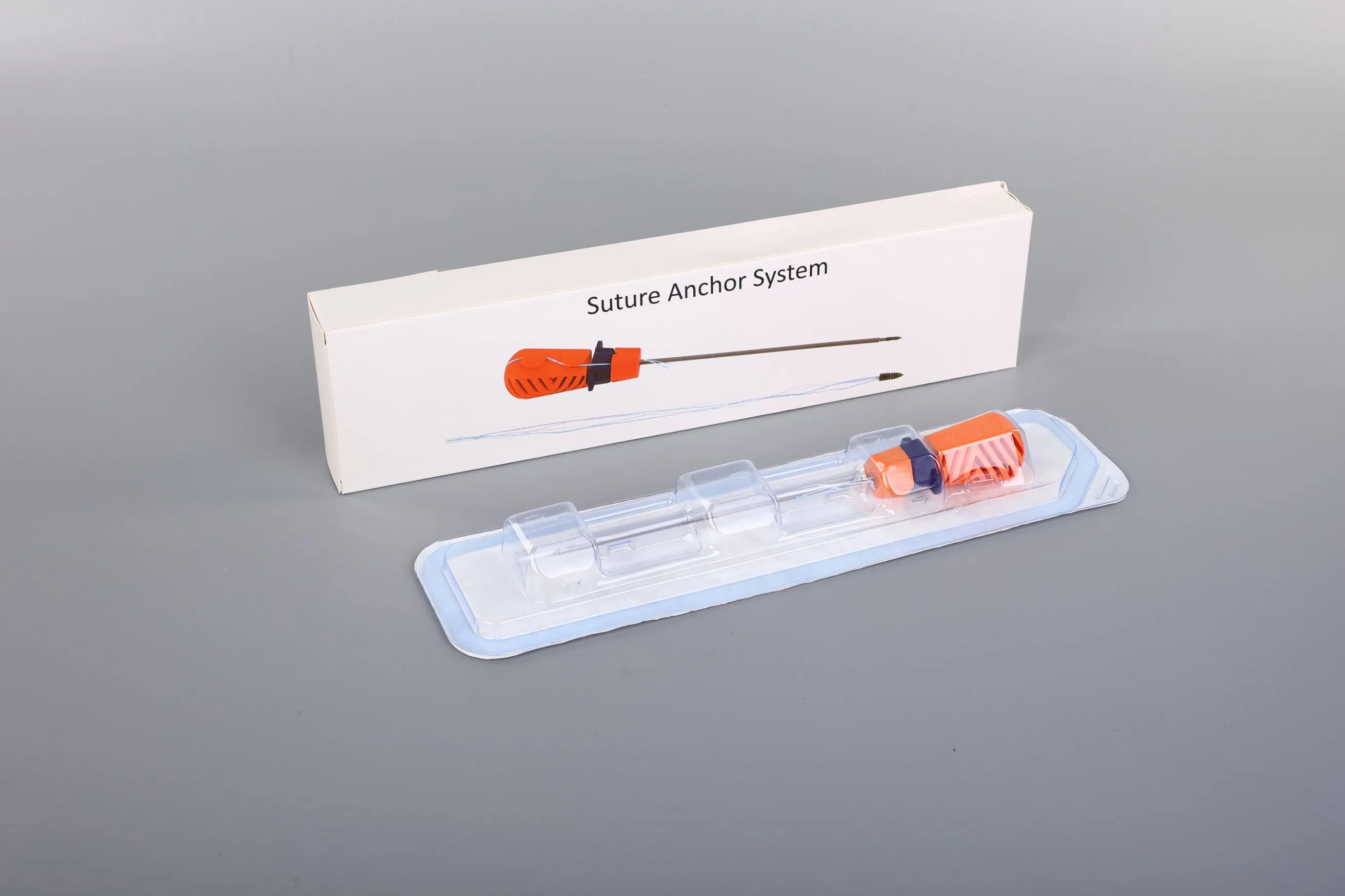
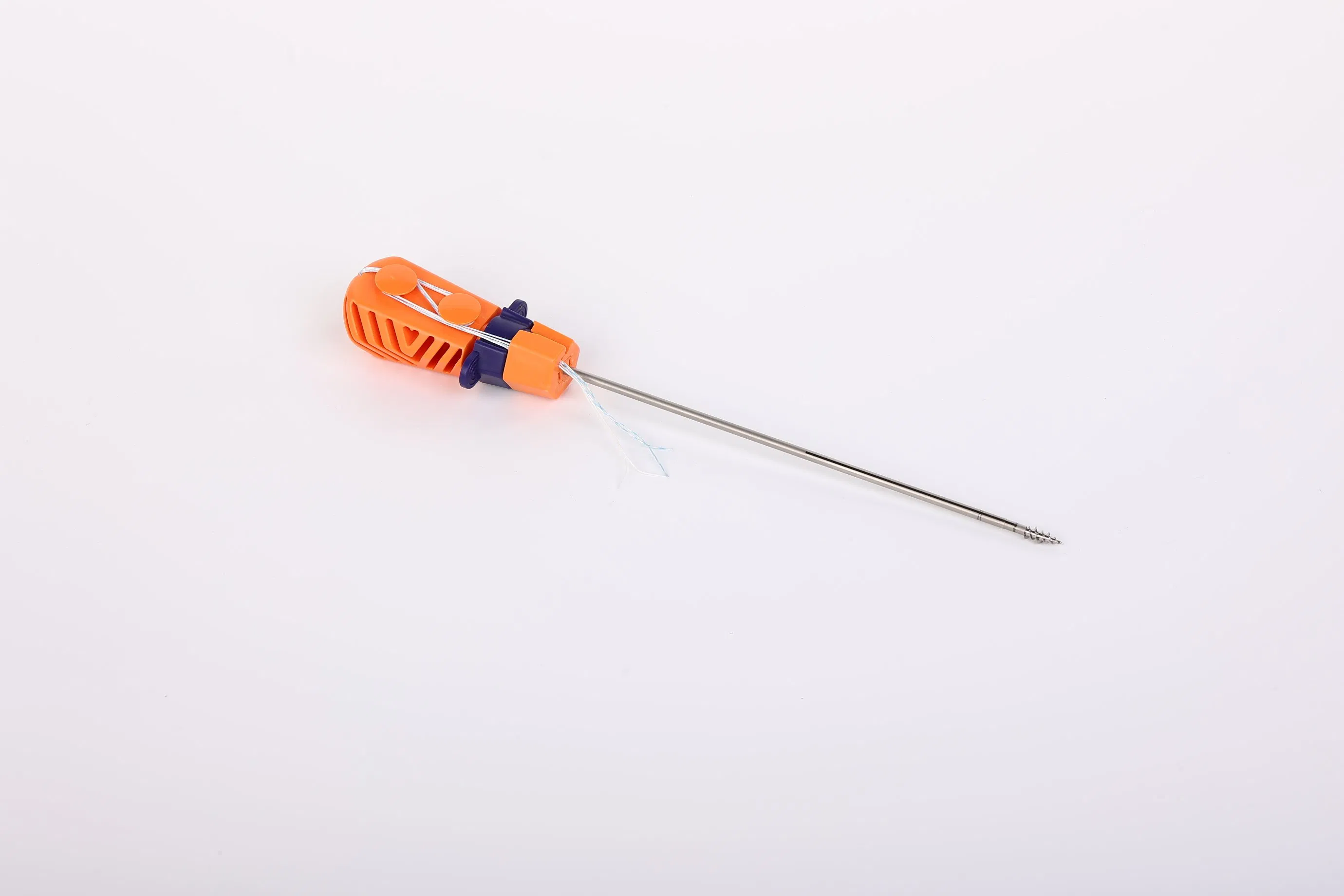
The Suture Anchor System consists of a high-performance anchor, non-absorbable suture, and a precision inserter. The anchor is engineered from Ti6Al4V alloy, fully meeting the stringent requirements of ISO 5832-3:2016 for orthopedic implants.
Our sutures are crafted from Ultra High Molecular Weight Polyethylene (UHMWPE) material. These are woven from multiple UHMWPE yarns without extra coating, complying with ASTM F2848-17 standards. For clear visibility during surgery, we offer blue sutures dyed with FDA-approved materials.

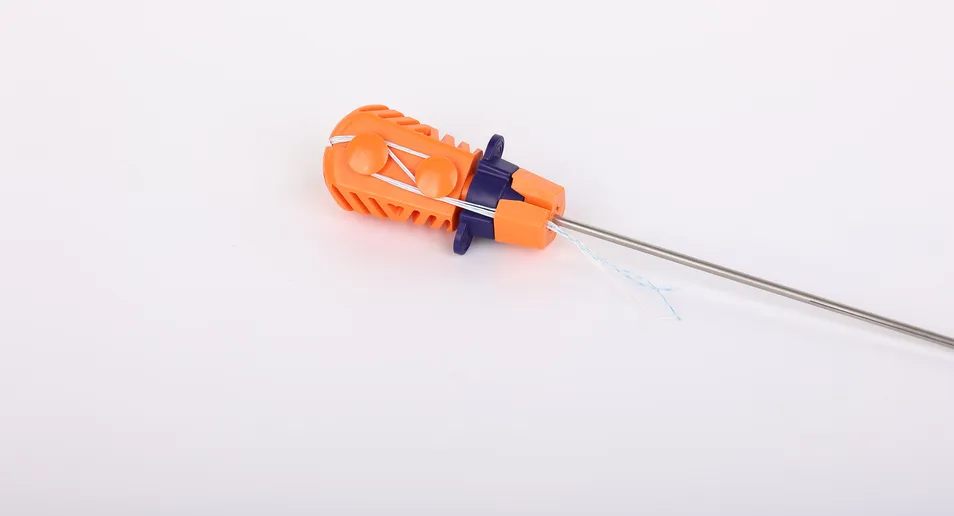

The instrument set includes an inserter where the parts contacting the human body are made of medical-grade stainless steel conforming to ASTM F899-12b. The handle is ergonomically designed using high-quality polycarbonate and ABS materials, ensuring stability and ease of use during orthopedic procedures.

Our production process involves advanced R&D and manufacturing capabilities. To guarantee the highest levels of safety and quality, all products are manufactured in Class 100,000 clean workshops and tested in Class 10,000 purification laboratories. Our orthopedic consumables are trusted in over 40 countries worldwide.


