

1 / 5
 Sete Orthopedic Implants
Sete Orthopedic Implants










| Model NO. | T1100-38 | Group | Adult |
|---|---|---|---|
| Delivery Time | Within 7 Days | Shipping | DHL, UPS, TNT, FedEx, Aramex, EMS |
| MOQ | 1 | OEM | Accepted |
| Package | PE Film | Origin | China |




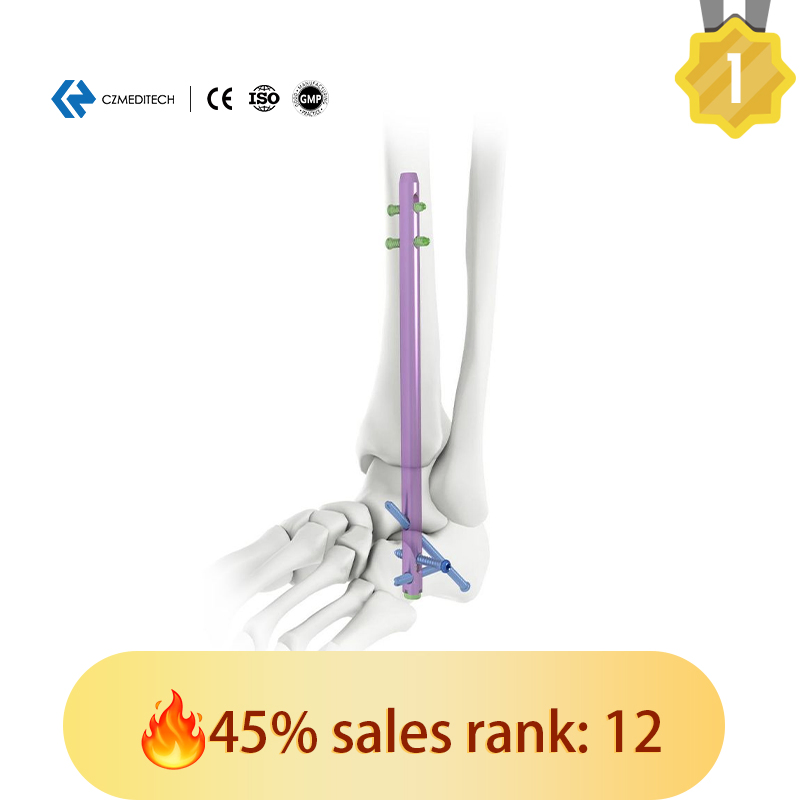
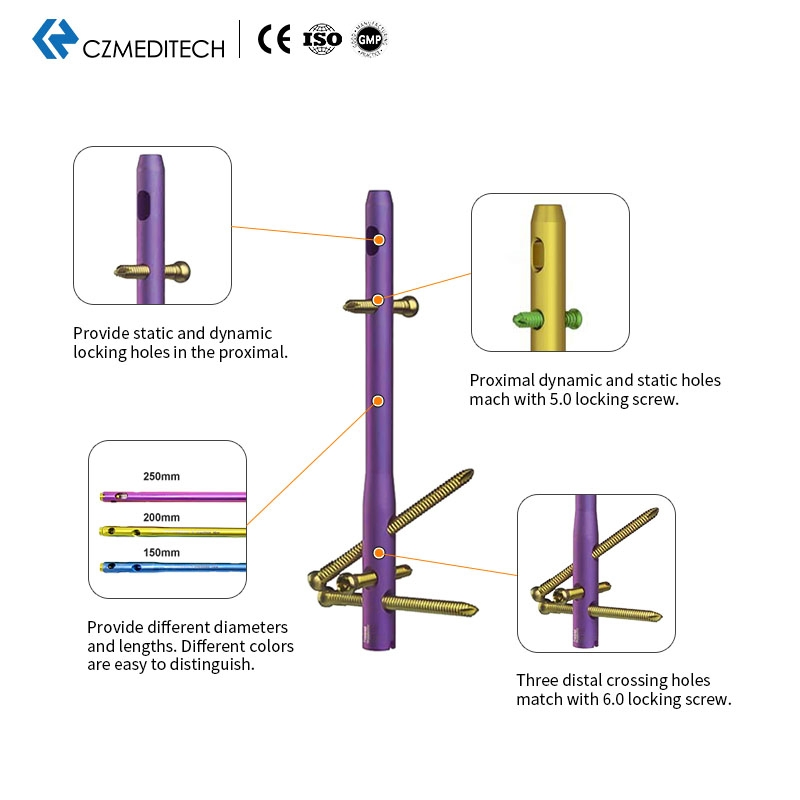
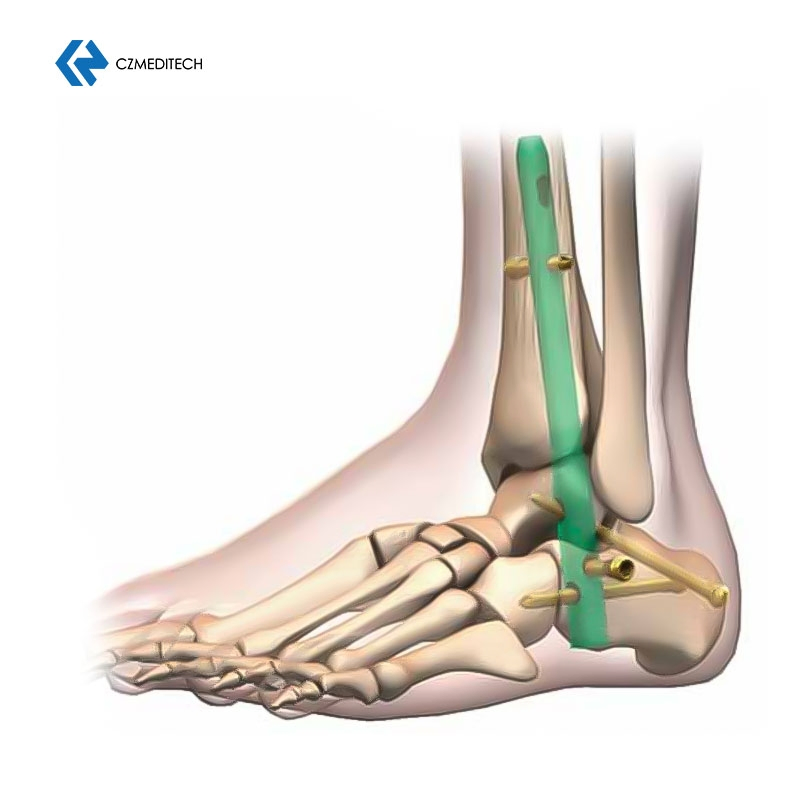
Size Variety: Different diameters and lengths are provided. Color-coded for easy identification.
Locking Holes: Features both static and dynamic locking holes in the proximal section.


Proximal Fixation: Dynamic and static holes match with 5.0mm locking screws.
Distal Fixation: Three distal crossing holes match with 6.0mm locking screws.
| REF | Description / Dimensions |
| Hindfoot Fusion Ankle Nail | 10*150/200/225/250 mm (L) |
| 10*150/200 mm (R) | |
| 11*150/200/250 mm (L) | |
| 11*150/200/225/250 mm (R) | |
| 12*150/200/250 mm (L) | |
| 12*150/200/250 mm (R) | |
| 5.0mm Locking Screws | 5.0*28mm to 60mm |
| 6.0mm Locking Screws | 6.0*26mm to 100mm |
Professional Orthopedic Solutions
Supplying to over 2,500+ clients across 70+ countries for over 15 years with extensive expertise.


Professional Support: Providing 1-year warranty and free repairs or replacements during the warranty period. Responses provided within 2 days for all queries.
CE Certified and ISO 13485 quality management system compliant.

ARAB HEALTH
UAE

MEDICA
Germany

HOSPITAL EXPO
Indonesia