



1 / 5
 Sete Orthopedic Implants
Sete Orthopedic Implants










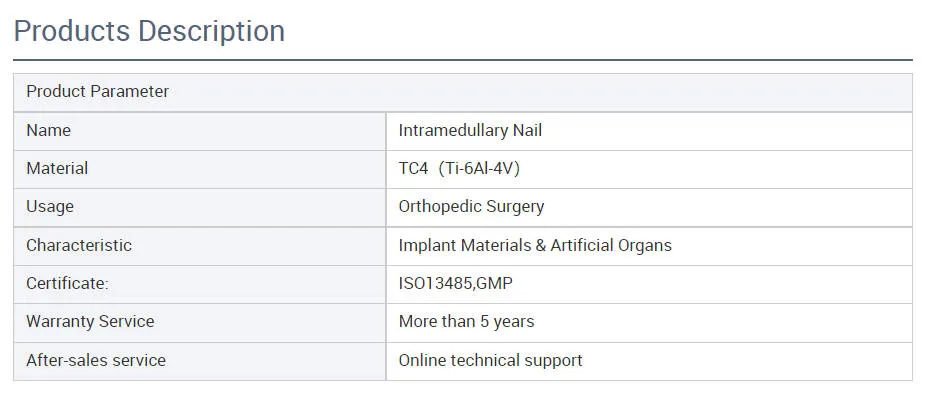


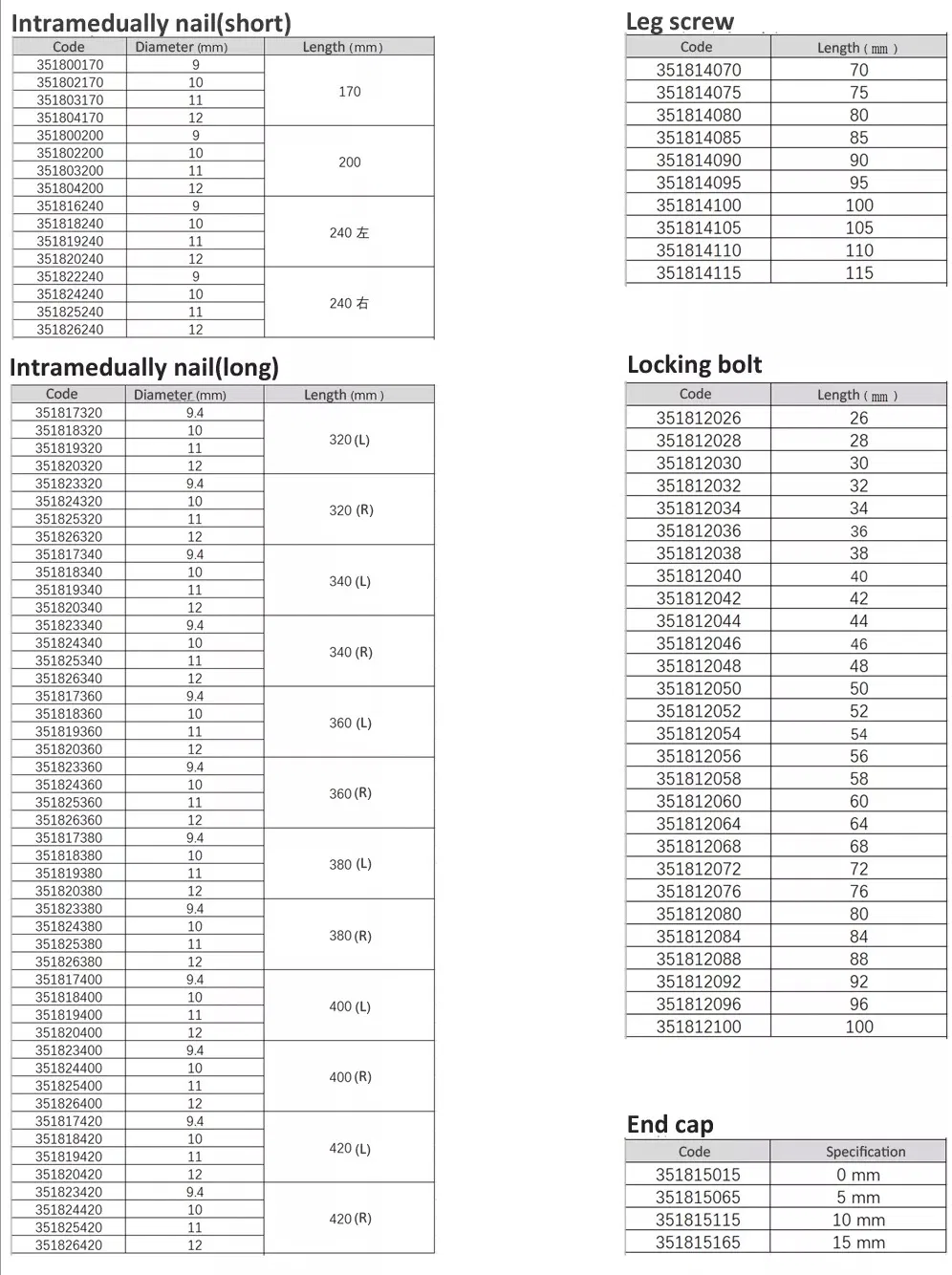
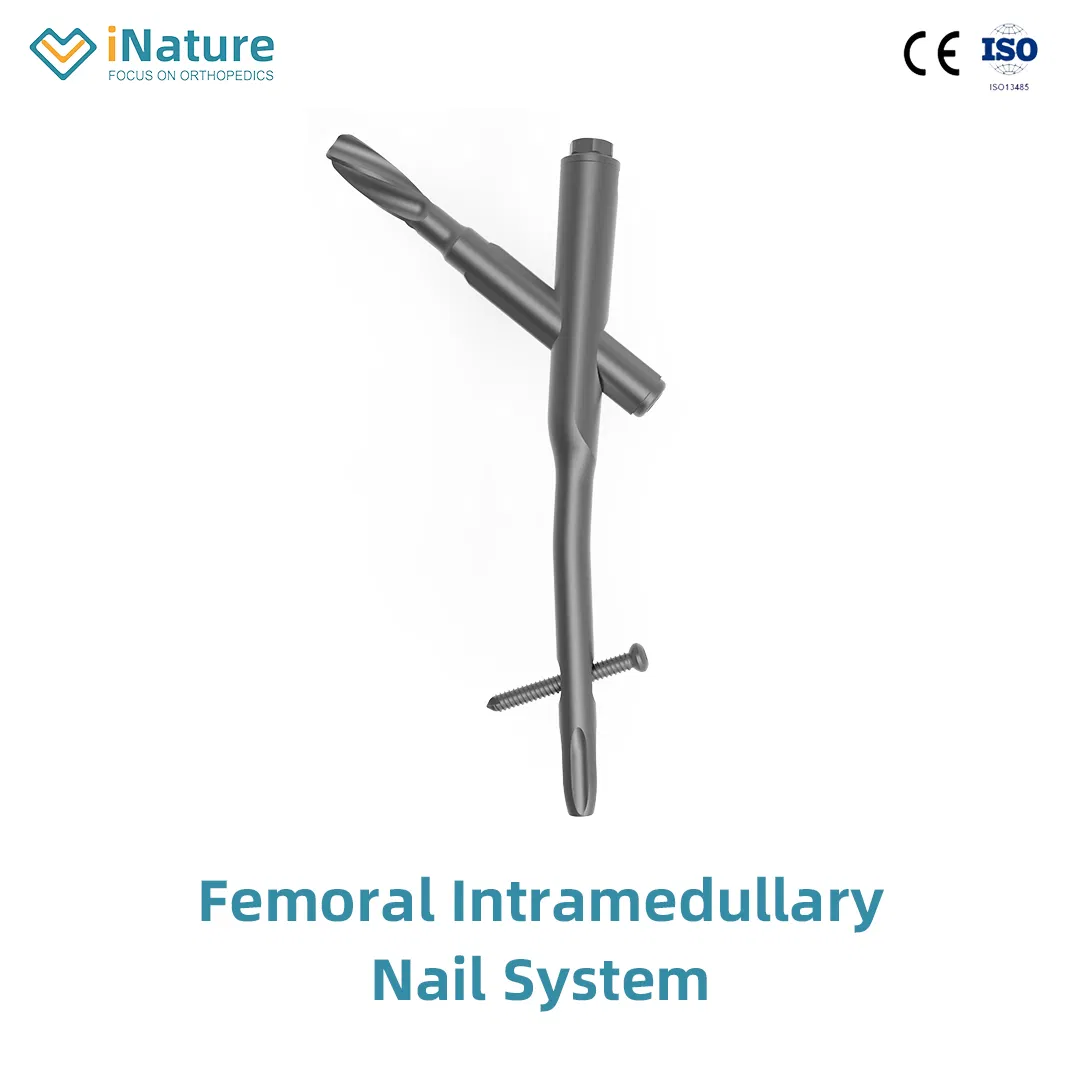
| ✔Material: Titanium |
| ✔Surface Finished: Oxidation / Milling for Titanium, Electrolysis for Stainless Steel |
| ⭐Key Features: |
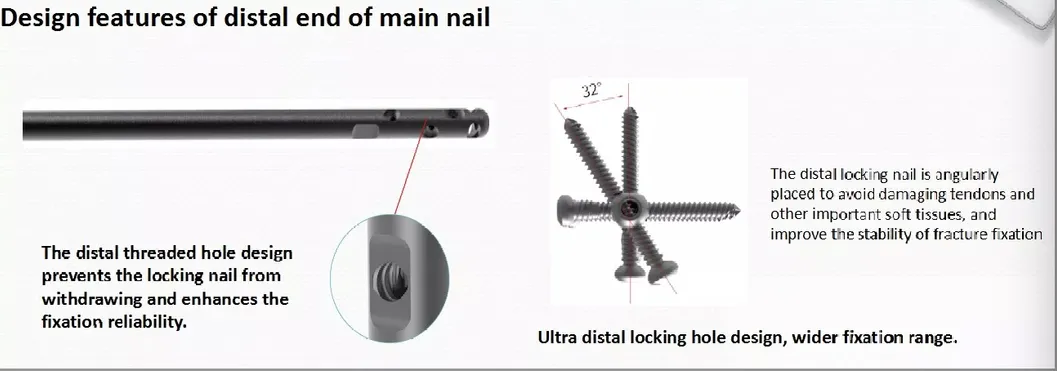
| • The medial-lateral angle of 5° allows insertion at the tip of the greater trochanter. |
| • Lateral flatten design eases insertion and reduces stress to the lateral cortical bone. |
| • Elastic groove tip design reduces concentration stress. |
| • 240mm distal curved design prevents nail tip contact with cortical bone, reducing pain. |
| • Lag screw insertion provides excellent compaction, ideal for osteoporotic patients. |
| • Anti-rotational stability achieved by a single element. |
| • Double thread locking bolt design to reduce operation time. |



